药明生物
提供端到端一站式解决方案
ADC分析和表征
Analytical Development and Characterization of ADCs and other Bioconjugates

生物偶联药结合了生物大分子(如蛋白质)和活性小分子,其结构往往相当复杂。所以它们的分析/表征成为重要环节。生物偶联研发部拥有支持ADC研发的优秀分析人员,多年的偶联物分析经验和尖端仪器设备。利用LC-MS,多种 HPLC等技术,BCD可以精确定量/表征偶联药物的DAR,残留小分子,内毒素等等。此外,生物偶联研发部也有很多仪器用于分析和表征。如利用cIEF等测定蛋白质偶联药物的更细致构型,MALS测定大分子质量分布,等等。
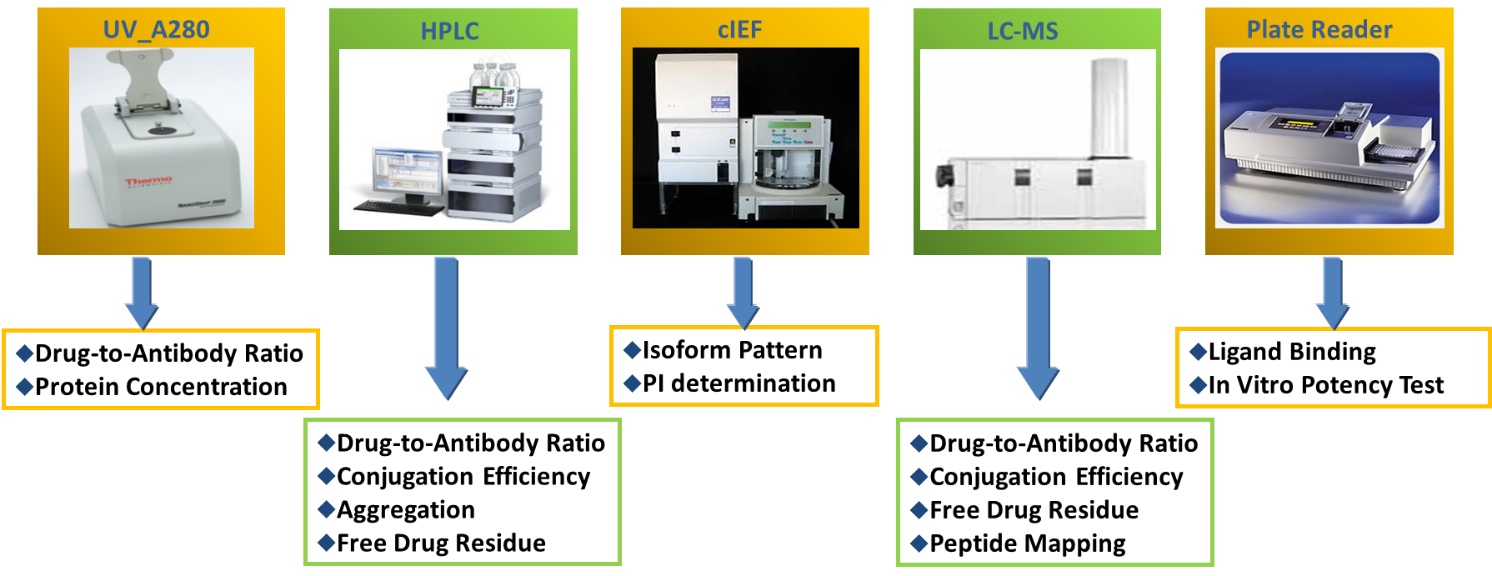
以下列举部分仪器及其分析项目:
- HPLC
-
- Drug-to-Antibody Ratio
- Conjugation Efficiency
- Aggregation
- Residue
- MALS
-
- Aggregation
- Nanoparticle Characterization
- LC-MS
-
- Drug-to-Antibody Ratio
- Conjugation Efficiency
- Free Drug Residue
- Peptide Mapping
- Plate Reader
-
- Ligand Binding/Affinity
- In vitro Potency Test
- Capillary Electrophoresis (CE) and Capillary Isoelectric Focusing (CIEF)
-
- Non-Reduced & Reduced (CE-SDS)
- Determination of pI (cIEF or iCIEF)
- UV/Vis (NanoDrop and SoloVPE®)
-
- Protein Concentration
- Drug-to-Antibody Ratio
其他生物偶联物表征能力:
- 毛细管电泳(电荷异质性和产品相关杂质)
- 天然MS(天然条件下完整分子量测定)
- 肽图(结合位点和占用率)
- 准确的Kd测定
更多参考信息
白皮书: The Watershed Moment for ADCs has Arrived
行业观点:
Antibody Drug Conjugate (ADC) Development And Manufacturing Challenges And Solutions
Webinar:
Overcoming Antibody-Drug Conjugate (ADC) Process Development









