- 关于我们
- 服务与能力
- 生产体系
- 哺乳动物细胞
$我们的专业能力覆盖从哺乳动物细胞培养到生物大分子的发现、开发与 cGMP 生产。依托 6 大新药发现平台、一流的 CMC 开发团队以及完善的供应链体系,我们能够为您的生物药开发提供全流程、一体化的解决方案。
- 哺乳动物细胞表达
$从概念到商业化,为您提供哺乳动物细胞生物药开发的一站式全流程服务。
哺乳动物细胞表达
从概念到商业化,为您提供哺乳动物细胞生物药开发的一站式全流程服务。
- 支持的产品类型:
- 单克隆抗体
$探索我们针对该类产品提供的全方位药物研发服务
单克隆抗体
探索我们针对该类产品提供的全方位药物研发服务
- 双特异性及多特异性抗体
$探索我们针对该类产品提供的全方位药物研发服务
双特异性及多特异性抗体
探索我们针对该类产品提供的全方位药物研发服务
- Fc融合蛋白
$探索我们针对该类产品提供的全方位药物研发服务
Fc融合蛋白
探索我们针对该类产品提供的全方位药物研发服务
- 抗体片段
$探索我们针对该类产品提供的全方位药物研发服务
抗体片段
探索我们针对该类产品提供的全方位药物研发服务
- 重组蛋白 / 酶 / 细胞因子
$探索我们针对该类产品提供的全方位药物研发服务
重组蛋白 / 酶 / 细胞因子
探索我们针对该类产品提供的全方位药物研发服务
- 抗体偶联药物(ADC)
$探索我们针对该类产品提供的全方位药物研发服务
抗体偶联药物(ADC)
探索我们针对该类产品提供的全方位药物研发服务
- 病毒样颗粒(VLP)
$探索我们针对该类产品提供的全方位药物研发服务
病毒样颗粒(VLP)
探索我们针对该类产品提供的全方位药物研发服务
- 微生物发酵
$全方位 CMC 开发与 cGMP 生产微生物发酵平台。提供基于大肠杆菌及酵母表达系统的质粒 DNA 与重组蛋白生产服务。
- 微生物发酵
$卓越品质,专为微生物发酵来源生物药提供专家级服务。
微生物发酵
卓越品质,专为微生物发酵来源生物药提供专家级服务。
- 支持的产品类型:
- 抗体片段
$探索我们针对该类产品提供的全方位药物研发服务
抗体片段
探索我们针对该类产品提供的全方位药物研发服务
- 酶
$探索我们针对该类产品提供的全方位药物研发服务
酶
探索我们针对该类产品提供的全方位药物研发服务
- 病毒样颗粒(VLP)
$探索我们针对该类产品提供的全方位药物研发服务
病毒样颗粒(VLP)
探索我们针对该类产品提供的全方位药物研发服务
- 核心能力
- 发现
$涵盖从早期概念至 IND 申报的集成化药物发现平台
- 研究探索
$药明生物提供行业专业知识、最先进的设施和多种抗体生成技术平台,用于发现新型单克隆 双特异性和多特异性抗体、免疫细胞因子和其他生物制剂。
研究探索
药明生物提供行业专业知识、最先进的设施和多种抗体生成技术平台,用于发现新型单克隆 双特异性和多特异性抗体、免疫细胞因子和其他生物制剂。
- 开发
$依托全球规模领先、经验丰富的开发团队,我们拥有卓越的资源、技术与专业实力,致力于以最高效、最具成本效益的方式,驱动您的项目顺利推进至 IND 与 BLA 申报。
- 细胞株工程
$无论是作为独立服务,还是作为我们集成化 CMC 开发平台的一部分,药明生物都能在广泛的生物药领域,为客户提供深厚的专业积淀,以及行业领先的细胞株工程与菌株开发周期。
细胞株工程
无论是作为独立服务,还是作为我们集成化 CMC 开发平台的一部分,药明生物都能在广泛的生物药领域,为客户提供深厚的专业积淀,以及行业领先的细胞株工程与菌株开发周期。
- 细胞株构建
- 分析科学
$我们提供全方位的分析检测服务,在过程控制(IPC)、成品放行及稳定性研究的方法开发领域拥有顶尖的专业实力。此外,我们还支持细胞株构建、工艺及制剂开发、产品表征、可开发性评估,以及其他支持 IND 和 BLA 申报的关键研究。
分析科学
我们提供全方位的分析检测服务,在过程控制(IPC)、成品放行及稳定性研究的方法开发领域拥有顶尖的专业实力。此外,我们还支持细胞株构建、工艺及制剂开发、产品表征、可开发性评估,以及其他支持 IND 和 BLA 申报的关键研究。
- 上下游工艺开发
$我们拥有多个上游与下游工艺开发实验室,支持分批补料、强化分批补料及连续生产工艺的建立与放大。我们的服务涵盖多种生物药类型,贯穿药物研发的早期及后期阶段。
上下游工艺开发
我们拥有多个上游与下游工艺开发实验室,支持分批补料、强化分批补料及连续生产工艺的建立与放大。我们的服务涵盖多种生物药类型,贯穿药物研发的早期及后期阶段。
- 细胞库建库
$我们提供一站式自有细胞库构建与细胞系表征服务,符合全球 GMP 法规及 ICH 指南要求;同时运营超过 20 个 cGMP 级细胞库车间,确保该关键 CMC 开发环节具备充足产能并可按时执行。
细胞库建库
我们提供一站式自有细胞库构建与细胞系表征服务,符合全球 GMP 法规及 ICH 指南要求;同时运营超过 20 个 cGMP 级细胞库车间,确保该关键 CMC 开发环节具备充足产能并可按时执行。
- 生产
$我们在四个国家布局了多座先进且高品质的 cGMP 生产设施,涵盖临床及商业化规模的药物原液(DS)和制剂(DP)生产,能够支持来源于哺乳动物及微生物表达系统的多种生物制品生产。
- 临床原液(DS)GMP生产
$运营多个高质量、先进的临床规模 cGMP 设施,用于生物制药原液(DS)生产,涵盖哺乳动物和微生物两种表达系统。
临床原液(DS)GMP生产
运营多个高质量、先进的临床规模 cGMP 设施,用于生物制药原液(DS)生产,涵盖哺乳动物和微生物两种表达系统。
- 临床制剂(DP)GMP生产
$多个高度灵活的临床规模制剂(DP)生产设施,按照全球监管机构定义的现行药品生产质量管理规范(cGMP)要求,用于生物制剂和注射用制剂的配方、灌装、贴标及包装。
临床制剂(DP)GMP生产
多个高度灵活的临床规模制剂(DP)生产设施,按照全球监管机构定义的现行药品生产质量管理规范(cGMP)要求,用于生物制剂和注射用制剂的配方、灌装、贴标及包装。
- 商业化生产
$药明生物在四个国家拥有多个先进的、高质量的cGMP原液和制剂生产设施。利用多种规格的、经过验证的西林瓶、胶塞和铝盖组合平台,能够在不同的临床阶段和商业化生产规模上进行水针或冻干制剂产品生产。
商业化生产
药明生物在四个国家拥有多个先进的、高质量的cGMP原液和制剂生产设施。利用多种规格的、经过验证的西林瓶、胶塞和铝盖组合平台,能够在不同的临床阶段和商业化生产规模上进行水针或冻干制剂产品生产。
- 药物生产
- 药物cGMP填充和完成
- 检测
$我们在工艺过程检测、产品表征、放行检测及稳定性方法的开发与检测方面具备深厚的专业能力,既可作为一体化生物药开发平台的支持服务提供,也可作为独立项目开展。我们覆盖广泛的分析检测与生物安全检测卓越中心,以及经监管机构批准的质量控制(QC)实验室,是我们为客户提供各项服务的核心支撑。
- 生物药安全检测
$我们拥有符合 EMA、ISO (CNAS) 及 CMA 认证的高质量自有生物安全检测设施,能够为原材料、细胞株及未加工原液提供外源因子筛查,并结合卓越的病毒清除验证能力,为客户提供一站式生物安全检测服务解决方案。
生物药安全检测
我们拥有符合 EMA、ISO (CNAS) 及 CMA 认证的高质量自有生物安全检测设施,能够为原材料、细胞株及未加工原液提供外源因子筛查,并结合卓越的病毒清除验证能力,为客户提供一站式生物安全检测服务解决方案。
- 分析检测
$凭借全方位的检测开发与分析测试能力,我们助力药物研发的全生命周期——从关键表征研究到支持 IND/BLA 申报的各项试验,包括专业的生物分析与法医鉴定。我们致力于为您独特的产品需求提供量身定制的定制化方案。
分析检测
凭借全方位的检测开发与分析测试能力,我们助力药物研发的全生命周期——从关键表征研究到支持 IND/BLA 申报的各项试验,包括专业的生物分析与法医鉴定。我们致力于为您独特的产品需求提供量身定制的定制化方案。
- 卓越中心
$我们的卓越中心(CoE)在产品全生命周期内提供专业的检测支持,旨在加速项目进程,并确保项目在商业化阶段具备完善的分析就绪能力。
卓越中心
我们的卓越中心(CoE)在产品全生命周期内提供专业的检测支持,旨在加速项目进程,并确保项目在商业化阶段具备完善的分析就绪能力。
- 质量标准
- 质量管理
$我们拥有世界一流的质量体系,并在全球各生产基地实现统一标准化管理。我们的质量体系已通过包括美国 FDA、欧洲 EMA、中国 NMPA、日本 PMDA、韩国 MFDS、新加坡 HSA、巴西 ANVISA 及加拿大卫生部(Health Canada)在内的多家全球监管机构认证,支持多种生物药品的生产与检测。
- 全球质量合规部(GQC)
$我们的全球质量与合规团队统筹审计、IT 质量及风险管控,将合规意识贯彻至每一个环节。这确保了我们交付的每一件生物制品都拥有卓越的安全性与疗效,并在执行标准上与您的要求高度对齐。
全球质量合规部(GQC)
我们的全球质量与合规团队统筹审计、IT 质量及风险管控,将合规意识贯彻至每一个环节。这确保了我们交付的每一件生物制品都拥有卓越的安全性与疗效,并在执行标准上与您的要求高度对齐。
- 质量保证
$全球合规体系、全员质量承诺。依托全球生产基地统一的 QA 标准,我们为生物药及疫苗的商业化生产提供稳定、可靠、符合国际主流监管要求的质量支撑。
质量保证
全球合规体系、全员质量承诺。依托全球生产基地统一的 QA 标准,我们为生物药及疫苗的商业化生产提供稳定、可靠、符合国际主流监管要求的质量支撑。
- 质量控制
$我们拥有符合法规要求的自有 QC 实验室,为所有临床及商业化 GMP 生产基地提供全流程支持。我们不仅确保生产前后的产品高质量检测,更对环境监测、清洗验证、仪器生命周期管理、样品/留样管理及审计等关键职能进行全面监督。
质量控制
我们拥有符合法规要求的自有 QC 实验室,为所有临床及商业化 GMP 生产基地提供全流程支持。我们不仅确保生产前后的产品高质量检测,更对环境监测、清洗验证、仪器生命周期管理、样品/留样管理及审计等关键职能进行全面监督。
- 法规事务
$依托深厚的法规专业积淀,我们为客户提供从 CMC 申报资料、药物递交到药物注册的全方位支持。自 2015 年起,我们已成功支持全球客户申报超过550 余项 IND、CTA、BLA、MAA、NDA 及 EUA 申报,完成200 多个 Module 3 CMC 申报件。
法规事务
依托深厚的法规专业积淀,我们为客户提供从 CMC 申报资料、药物递交到药物注册的全方位支持。自 2015 年起,我们已成功支持全球客户申报超过550 余项 IND、CTA、BLA、MAA、NDA 及 EUA 申报,完成200 多个 Module 3 CMC 申报件。
- 技术平台
- 发现
$药明生物提供了先进全面的抗体发现服务,用于创新型抗体的发现、鉴定和筛选提供全方位的服务。
- WuXiBody ® 双特异性抗体平台
$WuXiBody ® 平台是药明生物开发的一个创新的、专有的技术平台,用于扩大双特异性抗体应用范围。基于最新的工程设计,该平台可以加快6-18个月的研发进程,大幅度降低产品的成本。
WuXiBody ® 双特异性抗体平台
WuXiBody ® 平台是药明生物开发的一个创新的、专有的技术平台,用于扩大双特异性抗体应用范围。基于最新的工程设计,该平台可以加快6-18个月的研发进程,大幅度降低产品的成本。
- WuXiHYbrid™ 杂交瘤单克隆抗体研发平台
$WuXiHYbrid™ 是国内领先、世界一流杂交瘤抗体研发平台,突破性提高了抗体新药研发的质量和速度,已为国内外50+客户成功交付超过200个高质量的单克隆抗体研发项目。
WuXiHYbrid™ 杂交瘤单克隆抗体研发平台
WuXiHYbrid™ 是国内领先、世界一流杂交瘤抗体研发平台,突破性提高了抗体新药研发的质量和速度,已为国内外50+客户成功交付超过200个高质量的单克隆抗体研发项目。
- WuXiLiAb™ 噬菌体展示人抗体库
$全人天然抗体库选取60个健康供体, 总数大于6x10e9 的PBMC或CBMC, 每个供体都单独建库并系统QC,保证了文库的高质量和多样性。
WuXiLiAb™ 噬菌体展示人抗体库
全人天然抗体库选取60个健康供体, 总数大于6x10e9 的PBMC或CBMC, 每个供体都单独建库并系统QC,保证了文库的高质量和多样性。
- 开发
$前沿的生物工艺平台与技术,旨在以更快的速度、更高的效率和更具成本效益的方式,推动高质量生物制剂进入临床试验阶段。
- WuXian™ 定制化蛋白生产服务
$依托药明生物行业领先的高通量高表达、纯化和分析技术,提供各类蛋白生产服务,其中包括抗体、双抗、酶和重组蛋白表达。
WuXian™ 定制化蛋白生产服务
依托药明生物行业领先的高通量高表达、纯化和分析技术,提供各类蛋白生产服务,其中包括抗体、双抗、酶和重组蛋白表达。
- WuXia™ 细胞株构建
$药明生物为多种生物治疗药物提供全面的哺乳动物细胞系开发服务,从客户提供的DNA或蛋白质序列开始,到交付出高产量、高产品质量且稳定的单克隆结束。
WuXia™ 细胞株构建
药明生物为多种生物治疗药物提供全面的哺乳动物细胞系开发服务,从客户提供的DNA或蛋白质序列开始,到交付出高产量、高产品质量且稳定的单克隆结束。
- WuXiUPTM 超高效连续细胞培养生产平台
$WuXiUPTM 平台是强化型的灌流工艺,提供高产量,高质量的新一代生物药制造解决方案,灵活性高,成本低。
WuXiUPTM 超高效连续细胞培养生产平台
WuXiUPTM 平台是强化型的灌流工艺,提供高产量,高质量的新一代生物药制造解决方案,灵活性高,成本低。
- WuXiDARx™偶联技术平台
$药明合联研发了WuXiDARx™技术*,一种基于抗体天然半胱氨酸残基的偶联方式,为生物偶联药管线开发提供了更高的灵活性。
WuXiDARx™偶联技术平台
药明合联研发了WuXiDARx™技术*,一种基于抗体天然半胱氨酸残基的偶联方式,为生物偶联药管线开发提供了更高的灵活性。
- 生产
$先进的生物制造平台赋能全球医疗合作伙伴,助力生物药快速迈向临床并成功上市,造福全球患者。
- 一次性生物反应器
$我们运营着全球规模领先的数个一次性生物反应器生产基地。依托一次性使用系统在降低风险、生产灵活性、成本效益及环境友好性方面的卓越优势,为客户提供高效的生物药生产保障。
一次性生物反应器
我们运营着全球规模领先的数个一次性生物反应器生产基地。依托一次性使用系统在降低风险、生产灵活性、成本效益及环境友好性方面的卓越优势,为客户提供高效的生物药生产保障。
- Scale-Out生物药生产
$通过使用多个同等规模的反应器进行并联生产,我们能有效降低工艺放大风险,为您在临床至上市的各阶段提供极高的生产弹性,确保稳定供应。
Scale-Out生物药生产
通过使用多个同等规模的反应器进行并联生产,我们能有效降低工艺放大风险,为您在临床至上市的各阶段提供极高的生产弹性,确保稳定供应。
- 机器人无菌灌装
$我们采用全程序化、机器人自动化且无手套的隔离器技术,显著降低先进无菌制剂灌装过程中的生产风险。该系统具备极高的灵活性,能够支持各种复杂生物药及多样化包材系统的灌装需求。
机器人无菌灌装
我们采用全程序化、机器人自动化且无手套的隔离器技术,显著降低先进无菌制剂灌装过程中的生产风险。该系统具备极高的灵活性,能够支持各种复杂生物药及多样化包材系统的灌装需求。
- 连续细胞培养生产工艺
$结合强化灌流培养(IPC)与连续直接产物捕获(CDPC),并配备先进设备,实现生产效率提升并降低成本。
- Scale-Out生物药生产
概述
全球领先的开放式、一体化生物偶联制药能力和技术赋能平台 Watch our video to learn more about the WuXi XDC integrated services advantage
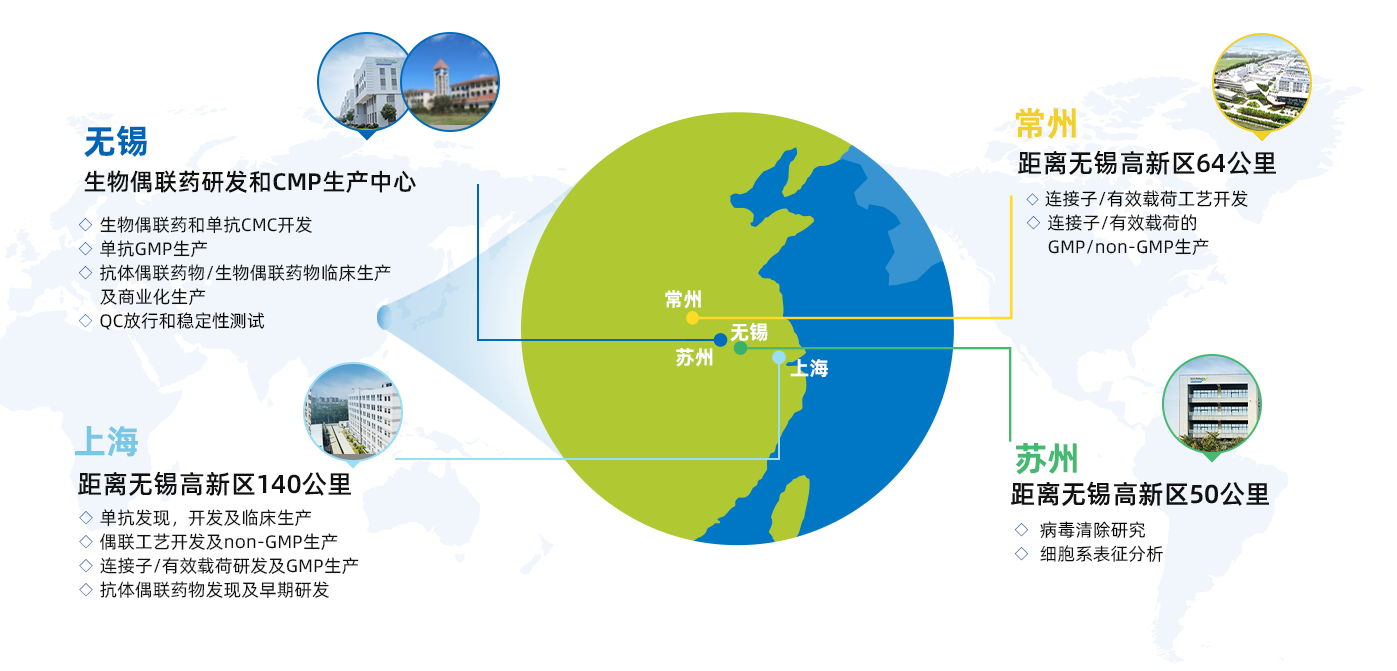
Watch our video to learn more about the WuXi XDC integrated services advantage药明合联(WuXi XDC)是由药明生物和合全药业联合成立的合资公司,是全球领先的开放式、一体化生物偶联制药能力和技术赋能平台。公司为全球生物偶联制药公司和生物偶联药开发者们提供全方位的端到端CRDMO服务,实现从概念到商业化生产的全过程整合,并通过高度集成ADC工艺研发和生产的供应链,加速并变革全球生物偶联药研发进程,为客户提供开放式,一体化的偶联制药技术平台,降低研发成本,造福病患。
药明合联已成功赋能多个ADC药物在15个月以内完成从DNA到新药临床试验申请(IND),相比传统开发时间几乎缩短了一半。截止2021年12月底,有60个处于CMC阶段的ADC及其他偶联药物在药明合联平台上开发,其中包括22个已提交IND的项目,6个项目进入后期临床试验阶段。
药明合联技术平台内的所有活动均在1-2小时车程范围内区域进行,从而提供了前所未有的以地理为中心的供应链和价值链。
药明合联主要由三个业务部门组成,上海生物偶联研发部负责偶联药物的早期研究和工艺开发,无锡高新区的生物偶联药园区负责偶联药物的GMP偶联生产,成品制剂及工艺开发和制剂生产,常州小分子基地承担载荷和连接子的研发和生产。
更多参考信息
白皮书: The Watershed Moment for ADCs has Arrived
行业观点:
Antibody Drug Conjugate (ADC) Development And Manufacturing Challenges And Solutions
Webinar:
Overcoming Antibody-Drug Conjugate (ADC) Process Development
抗体偶联药物新闻
- WuXia™ 细胞株构建
- WuXiHYbrid™ 杂交瘤单克隆抗体研发平台
- WuXiBody ® 双特异性抗体平台
- 质量保证
- 全球质量合规部(GQC)
- 分析检测
- 临床制剂(DP)GMP生产
- 开发
- 研究探索
- 哺乳动物细胞表达
- 哺乳动物细胞
- 生产体系
WuXi Biologics
Offering End-to-End Solutions
- 关于我们
- 服务与能力
- 生产体系
- 哺乳动物细胞
$我们的专业能力覆盖从哺乳动物细胞培养到生物大分子的发现、开发与 cGMP 生产。依托 6 大新药发现平台、一流的 CMC 开发团队以及完善的供应链体系,我们能够为您的生物药开发提供全流程、一体化的解决方案。

- 返回
- 哺乳动物细胞表达
$从概念到商业化,为您提供哺乳动物细胞生物药开发的一站式全流程服务。
哺乳动物细胞表达
从概念到商业化,为您提供哺乳动物细胞生物药开发的一站式全流程服务。
- 支持的产品类型:
- 单克隆抗体
$探索我们针对该类产品提供的全方位药物研发服务
单克隆抗体
探索我们针对该类产品提供的全方位药物研发服务
- 双特异性及多特异性抗体
$探索我们针对该类产品提供的全方位药物研发服务
双特异性及多特异性抗体
探索我们针对该类产品提供的全方位药物研发服务
- Fc融合蛋白
$探索我们针对该类产品提供的全方位药物研发服务
Fc融合蛋白
探索我们针对该类产品提供的全方位药物研发服务
- 抗体片段
$探索我们针对该类产品提供的全方位药物研发服务
抗体片段
探索我们针对该类产品提供的全方位药物研发服务
- 重组蛋白 / 酶 / 细胞因子
$探索我们针对该类产品提供的全方位药物研发服务
重组蛋白 / 酶 / 细胞因子
探索我们针对该类产品提供的全方位药物研发服务
- 抗体偶联药物(ADC)
$探索我们针对该类产品提供的全方位药物研发服务
- 微生物发酵
$全方位 CMC 开发与 cGMP 生产微生物发酵平台。提供基于大肠杆菌及酵母表达系统的质粒 DNA 与重组蛋白生产服务。

- 返回
- 微生物发酵
$卓越品质,专为微生物发酵来源生物药提供专家级服务。
微生物发酵
卓越品质,专为微生物发酵来源生物药提供专家级服务。
- 支持的产品类型:
- 抗体片段
$探索我们针对该类产品提供的全方位药物研发服务
抗体片段
探索我们针对该类产品提供的全方位药物研发服务
- 酶
$探索我们针对该类产品提供的全方位药物研发服务
- 核心能力
- 发现
$涵盖从早期概念至 IND 申报的集成化药物发现平台

- 开发
$依托全球规模领先、经验丰富的开发团队,我们拥有卓越的资源、技术与专业实力,致力于以最高效、最具成本效益的方式,驱动您的项目顺利推进至 IND 与 BLA 申报。

- 返回
- 细胞株工程
$无论是作为独立服务,还是作为我们集成化 CMC 开发平台的一部分,药明生物都能在广泛的生物药领域,为客户提供深厚的专业积淀,以及行业领先的细胞株工程与菌株开发周期。
- 细胞株构建
- 分析科学
$我们提供全方位的分析检测服务,在过程控制(IPC)、成品放行及稳定性研究的方法开发领域拥有顶尖的专业实力。此外,我们还支持细胞株构建、工艺及制剂开发、产品表征、可开发性评估,以及其他支持 IND 和 BLA 申报的关键研究。
分析科学
我们提供全方位的分析检测服务,在过程控制(IPC)、成品放行及稳定性研究的方法开发领域拥有顶尖的专业实力。此外,我们还支持细胞株构建、工艺及制剂开发、产品表征、可开发性评估,以及其他支持 IND 和 BLA 申报的关键研究。
- 上下游工艺开发
$我们拥有多个上游与下游工艺开发实验室,支持分批补料、强化分批补料及连续生产工艺的建立与放大。我们的服务涵盖多种生物药类型,贯穿药物研发的早期及后期阶段。
上下游工艺开发
我们拥有多个上游与下游工艺开发实验室,支持分批补料、强化分批补料及连续生产工艺的建立与放大。我们的服务涵盖多种生物药类型,贯穿药物研发的早期及后期阶段。
- 细胞库建库
$我们提供一站式自有细胞库构建与细胞系表征服务,符合全球 GMP 法规及 ICH 指南要求;同时运营超过 20 个 cGMP 级细胞库车间,确保该关键 CMC 开发环节具备充足产能并可按时执行。
细胞库建库
我们提供一站式自有细胞库构建与细胞系表征服务,符合全球 GMP 法规及 ICH 指南要求;同时运营超过 20 个 cGMP 级细胞库车间,确保该关键 CMC 开发环节具备充足产能并可按时执行。
- 生产
$我们在四个国家布局了多座先进且高品质的 cGMP 生产设施,涵盖临床及商业化规模的药物原液(DS)和制剂(DP)生产,能够支持来源于哺乳动物及微生物表达系统的多种生物制品生产。

- 返回
- 临床原液(DS)GMP生产
$运营多个高质量、先进的临床规模 cGMP 设施,用于生物制药原液(DS)生产,涵盖哺乳动物和微生物两种表达系统。
临床原液(DS)GMP生产
运营多个高质量、先进的临床规模 cGMP 设施,用于生物制药原液(DS)生产,涵盖哺乳动物和微生物两种表达系统。
- 临床制剂(DP)GMP生产
$多个高度灵活的临床规模制剂(DP)生产设施,按照全球监管机构定义的现行药品生产质量管理规范(cGMP)要求,用于生物制剂和注射用制剂的配方、灌装、贴标及包装。
临床制剂(DP)GMP生产
多个高度灵活的临床规模制剂(DP)生产设施,按照全球监管机构定义的现行药品生产质量管理规范(cGMP)要求,用于生物制剂和注射用制剂的配方、灌装、贴标及包装。
- 商业化生产
$药明生物在四个国家拥有多个先进的、高质量的cGMP原液和制剂生产设施。利用多种规格的、经过验证的西林瓶、胶塞和铝盖组合平台,能够在不同的临床阶段和商业化生产规模上进行水针或冻干制剂产品生产。
商业化生产
药明生物在四个国家拥有多个先进的、高质量的cGMP原液和制剂生产设施。利用多种规格的、经过验证的西林瓶、胶塞和铝盖组合平台,能够在不同的临床阶段和商业化生产规模上进行水针或冻干制剂产品生产。
- 药物生产
- 药物cGMP填充和完成
- 临床制剂(DP)GMP生产
- 检测
$我们在工艺过程检测、产品表征、放行检测及稳定性方法的开发与检测方面具备深厚的专业能力,既可作为一体化生物药开发平台的支持服务提供,也可作为独立项目开展。我们覆盖广泛的分析检测与生物安全检测卓越中心,以及经监管机构批准的质量控制(QC)实验室,是我们为客户提供各项服务的核心支撑。

- 返回
- 生物药安全检测
$我们拥有符合 EMA、ISO (CNAS) 及 CMA 认证的高质量自有生物安全检测设施,能够为原材料、细胞株及未加工原液提供外源因子筛查,并结合卓越的病毒清除验证能力,为客户提供一站式生物安全检测服务解决方案。
生物药安全检测
我们拥有符合 EMA、ISO (CNAS) 及 CMA 认证的高质量自有生物安全检测设施,能够为原材料、细胞株及未加工原液提供外源因子筛查,并结合卓越的病毒清除验证能力,为客户提供一站式生物安全检测服务解决方案。
- 分析检测
$凭借全方位的检测开发与分析测试能力,我们助力药物研发的全生命周期——从关键表征研究到支持 IND/BLA 申报的各项试验,包括专业的生物分析与法医鉴定。我们致力于为您独特的产品需求提供量身定制的定制化方案。
分析检测
凭借全方位的检测开发与分析测试能力,我们助力药物研发的全生命周期——从关键表征研究到支持 IND/BLA 申报的各项试验,包括专业的生物分析与法医鉴定。我们致力于为您独特的产品需求提供量身定制的定制化方案。
- 卓越中心
$我们的卓越中心(CoE)在产品全生命周期内提供专业的检测支持,旨在加速项目进程,并确保项目在商业化阶段具备完善的分析就绪能力。
- 质量标准
- 质量管理
$我们拥有世界一流的质量体系,并在全球各生产基地实现统一标准化管理。我们的质量体系已通过包括美国 FDA、欧洲 EMA、中国 NMPA、日本 PMDA、韩国 MFDS、新加坡 HSA、巴西 ANVISA 及加拿大卫生部(Health Canada)在内的多家全球监管机构认证,支持多种生物药品的生产与检测。

- 返回
- 全球质量合规部(GQC)
$我们的全球质量与合规团队统筹审计、IT 质量及风险管控,将合规意识贯彻至每一个环节。这确保了我们交付的每一件生物制品都拥有卓越的安全性与疗效,并在执行标准上与您的要求高度对齐。
全球质量合规部(GQC)
我们的全球质量与合规团队统筹审计、IT 质量及风险管控,将合规意识贯彻至每一个环节。这确保了我们交付的每一件生物制品都拥有卓越的安全性与疗效,并在执行标准上与您的要求高度对齐。
- 质量保证
$全球合规体系、全员质量承诺。依托全球生产基地统一的 QA 标准,我们为生物药及疫苗的商业化生产提供稳定、可靠、符合国际主流监管要求的质量支撑。
质量保证
全球合规体系、全员质量承诺。依托全球生产基地统一的 QA 标准,我们为生物药及疫苗的商业化生产提供稳定、可靠、符合国际主流监管要求的质量支撑。
- 质量控制
$我们拥有符合法规要求的自有 QC 实验室,为所有临床及商业化 GMP 生产基地提供全流程支持。我们不仅确保生产前后的产品高质量检测,更对环境监测、清洗验证、仪器生命周期管理、样品/留样管理及审计等关键职能进行全面监督。
质量控制
我们拥有符合法规要求的自有 QC 实验室,为所有临床及商业化 GMP 生产基地提供全流程支持。我们不仅确保生产前后的产品高质量检测,更对环境监测、清洗验证、仪器生命周期管理、样品/留样管理及审计等关键职能进行全面监督。
- 法规事务
$依托深厚的法规专业积淀,我们为客户提供从 CMC 申报资料、药物递交到药物注册的全方位支持。自 2015 年起,我们已成功支持全球客户申报超过550 余项 IND、CTA、BLA、MAA、NDA 及 EUA 申报,完成200 多个 Module 3 CMC 申报件。
法规事务
依托深厚的法规专业积淀,我们为客户提供从 CMC 申报资料、药物递交到药物注册的全方位支持。自 2015 年起,我们已成功支持全球客户申报超过550 余项 IND、CTA、BLA、MAA、NDA 及 EUA 申报,完成200 多个 Module 3 CMC 申报件。
- 技术平台
- 发现
$药明生物提供了先进全面的抗体发现服务,用于创新型抗体的发现、鉴定和筛选提供全方位的服务。

- 返回
- WuXiBody ® 双特异性抗体平台
$WuXiBody ® 平台是药明生物开发的一个创新的、专有的技术平台,用于扩大双特异性抗体应用范围。基于最新的工程设计,该平台可以加快6-18个月的研发进程,大幅度降低产品的成本。
WuXiBody ® 双特异性抗体平台
WuXiBody ® 平台是药明生物开发的一个创新的、专有的技术平台,用于扩大双特异性抗体应用范围。基于最新的工程设计,该平台可以加快6-18个月的研发进程,大幅度降低产品的成本。
- WuXiHYbrid™ 杂交瘤单克隆抗体研发平台
$WuXiHYbrid™ 是国内领先、世界一流杂交瘤抗体研发平台,突破性提高了抗体新药研发的质量和速度,已为国内外50+客户成功交付超过200个高质量的单克隆抗体研发项目。
WuXiHYbrid™ 杂交瘤单克隆抗体研发平台
WuXiHYbrid™ 是国内领先、世界一流杂交瘤抗体研发平台,突破性提高了抗体新药研发的质量和速度,已为国内外50+客户成功交付超过200个高质量的单克隆抗体研发项目。
- WuXiLiAb™ 噬菌体展示人抗体库
$全人天然抗体库选取60个健康供体, 总数大于6x10e9 的PBMC或CBMC, 每个供体都单独建库并系统QC,保证了文库的高质量和多样性。
- 开发
$前沿的生物工艺平台与技术,旨在以更快的速度、更高的效率和更具成本效益的方式,推动高质量生物制剂进入临床试验阶段。

- 返回
- WuXian™ 定制化蛋白生产服务
$依托药明生物行业领先的高通量高表达、纯化和分析技术,提供各类蛋白生产服务,其中包括抗体、双抗、酶和重组蛋白表达。
WuXian™ 定制化蛋白生产服务
依托药明生物行业领先的高通量高表达、纯化和分析技术,提供各类蛋白生产服务,其中包括抗体、双抗、酶和重组蛋白表达。
- WuXia™ 细胞株构建
$药明生物为多种生物治疗药物提供全面的哺乳动物细胞系开发服务,从客户提供的DNA或蛋白质序列开始,到交付出高产量、高产品质量且稳定的单克隆结束。
WuXia™ 细胞株构建
药明生物为多种生物治疗药物提供全面的哺乳动物细胞系开发服务,从客户提供的DNA或蛋白质序列开始,到交付出高产量、高产品质量且稳定的单克隆结束。
- WuXiUPTM 超高效连续细胞培养生产平台
$WuXiUPTM 平台是强化型的灌流工艺,提供高产量,高质量的新一代生物药制造解决方案,灵活性高,成本低。
WuXiUPTM 超高效连续细胞培养生产平台
WuXiUPTM 平台是强化型的灌流工艺,提供高产量,高质量的新一代生物药制造解决方案,灵活性高,成本低。
- WuXiDARx™偶联技术平台
$药明合联研发了WuXiDARx™技术*,一种基于抗体天然半胱氨酸残基的偶联方式,为生物偶联药管线开发提供了更高的灵活性。
- 生产
$先进的生物制造平台赋能全球医疗合作伙伴,助力生物药快速迈向临床并成功上市,造福全球患者。
菜单
- 返回
- 一次性生物反应器
$我们运营着全球规模领先的数个一次性生物反应器生产基地。依托一次性使用系统在降低风险、生产灵活性、成本效益及环境友好性方面的卓越优势,为客户提供高效的生物药生产保障。
一次性生物反应器
我们运营着全球规模领先的数个一次性生物反应器生产基地。依托一次性使用系统在降低风险、生产灵活性、成本效益及环境友好性方面的卓越优势,为客户提供高效的生物药生产保障。
- Scale-Out生物药生产
$通过使用多个同等规模的反应器进行并联生产,我们能有效降低工艺放大风险,为您在临床至上市的各阶段提供极高的生产弹性,确保稳定供应。
Scale-Out生物药生产
通过使用多个同等规模的反应器进行并联生产,我们能有效降低工艺放大风险,为您在临床至上市的各阶段提供极高的生产弹性,确保稳定供应。
- 机器人无菌灌装
$我们采用全程序化、机器人自动化且无手套的隔离器技术,显著降低先进无菌制剂灌装过程中的生产风险。该系统具备极高的灵活性,能够支持各种复杂生物药及多样化包材系统的灌装需求。
机器人无菌灌装
我们采用全程序化、机器人自动化且无手套的隔离器技术,显著降低先进无菌制剂灌装过程中的生产风险。该系统具备极高的灵活性,能够支持各种复杂生物药及多样化包材系统的灌装需求。
- 连续细胞培养生产工艺
$结合强化灌流培养(IPC)与连续直接产物捕获(CDPC),并配备先进设备,实现生产效率提升并降低成本。
- Scale-Out生物药生产
- 可持续发展
- 职业发展
- 投资者
- 新闻与媒体
- WuXia™ 细胞株构建
- WuXiHYbrid™ 杂交瘤单克隆抗体研发平台
- 质量保证
- 分析检测
- 上下游工艺开发
- 开发
- 哺乳动物细胞
- 生产体系







